|
5/15/2023 0 Comments Atoms join together to form  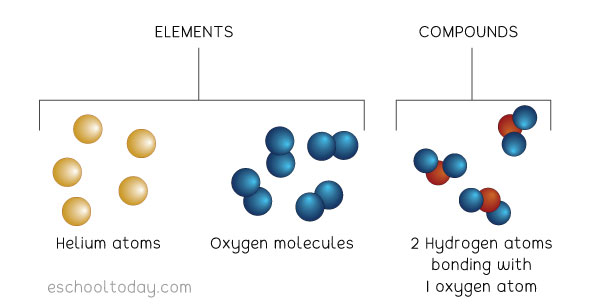
It is worth noticing here that all the valencies of all the atoms in both the molecules are fully satisfied and hence there is no further scope of bonding.Ī ‘complex’ situation arises when one comes to know that the molecule CoCl 3 can encompass six ammonia molecules, resulting into a third independent entity. Thus an ammonia molecule has three N H covalent bonds and one lone pair of electrons over the nitrogen atom. Keeping an electron pair in one of these orbitals while the other three remains half filled, an opportunity for three hydrogen atoms to contribute one electron each for the formation of a covalent bond with nitrogen, can also be explained. 
Similarly, the valence shell ( n = 2) of nitrogen (N = 7) contains five electrons and four orbitals (2s, 2p x, 2p y and 2p z). For instance, the +3 formal oxidation of cobalt in cobalt chloride is balanced by three uni-negative chloride ions and the coexistence of these ionic moieties to form a molecule is understood and explained. The bonding in compounds like CoCl 3 and NH 3 were easily understood and explained and hence such compounds were regarded as simple compounds. It is worth noting that the electron was discovered subsequent to Werner's theory. In the events, Werner's coordination theory (1893) became the base of the modern coordination chemistry. Thus, before the coordination chemistry was structured, the coordination compounds, complexes and chelates found their applications.Ī systematic investigation of structure and bonding in coordination chemistry began with the inquisitiveness of Tassaert (1798), which was extended by distinguished chemists like Wilhelm Blomstrand, Jorgensen and Alfred Werner until the end of the nineteenth century. A platinum complex K 2 offered an application for the refinement of platinum metal.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
